 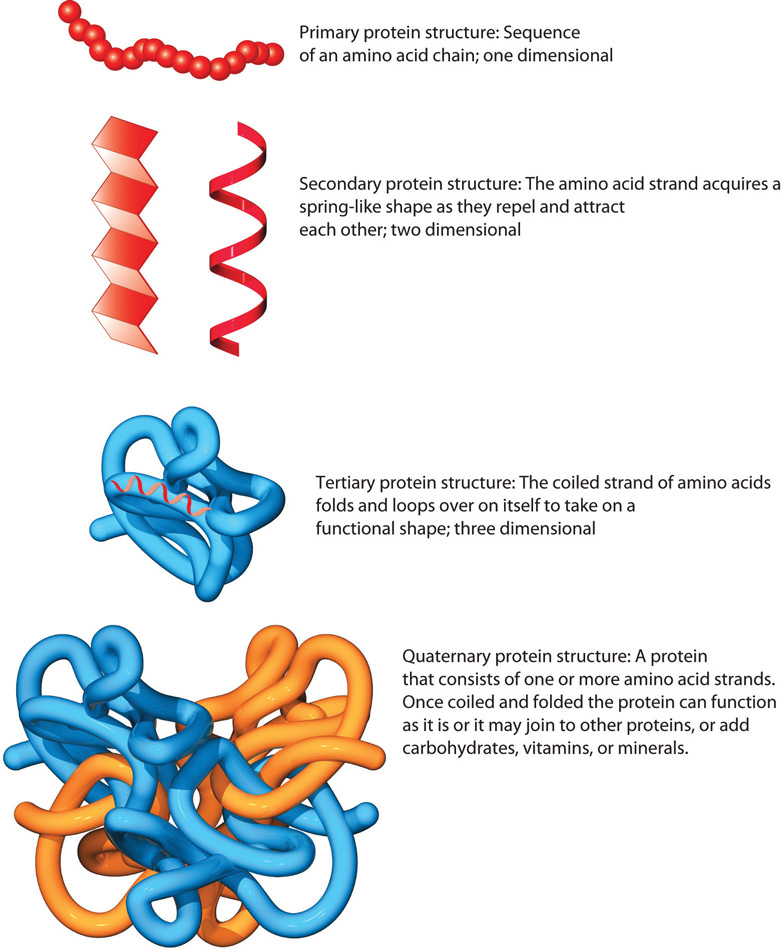
When a protein loses its three-dimensional shape, it may no longer be functional. These include hydrophobic interactions, ionic bonding, hydrogen bonding and disulfide linkages.Īll of these interactions, weak and strong, determine the final three-dimensional shape of the protein. The tertiary structure of proteins is determined by a variety of chemical interactions. Interaction between cysteine side chains forms disulfide linkages in the presence of oxygen, the only covalent bond forming during protein folding.įigure 5. The former types of interactions are also known as hydrophobic interactions. When protein folding takes place, the hydrophobic R groups of nonpolar amino acids lay in the interior of the protein, whereas the hydrophilic R groups lay on the outside. For example, R groups with like charges are repelled by each other and those with unlike charges are attracted to each other (ionic bonds). The nature of the R groups found in the amino acids involved can counteract the formation of the hydrogen bonds described for standard secondary structures. Primarily, the interactions among R groups creates the complex three-dimensional tertiary structure of a protein. This structure is in part due to chemical interactions at work on the polypeptide chain. The unique three-dimensional structure of a polypeptide is its tertiary structure (Figure 5). The α-helix and β-pleated sheet structures are found in most globular and fibrous proteins and they play an important structural role. The pleated segments align parallel or antiparallel to each other, and hydrogen bonds form between the partially positive nitrogen atom in the amino group and the partially negative oxygen atom in the carbonyl group of the peptide backbone. The R groups are attached to the carbons and extend above and below the folds of the pleat. In the β-pleated sheet, the “pleats” are formed by hydrogen bonding between atoms on the backbone of the polypeptide chain. The R groups (the variant groups) of the polypeptide protrude out from the α-helix chain. Certain amino acids have a propensity to form an α-helix, while others have a propensity to form a β-pleated sheet.Įvery helical turn in an alpha helix has 3.6 amino acid residues. 
The α-helix and β-pleated sheet are secondary structures of proteins that form because of hydrogen bonding between carbonyl and amino groups in the peptide backbone. The hydrogen bonds form between the oxygen atom in the carbonyl group in one amino acid and another amino acid that is four amino acids farther along the chain.įigure 4. Both structures are the α-helix structure-the helix held in shape by hydrogen bonds. The most common are the α-helix and β-pleated sheet structures (Figure 4). The local folding of the polypeptide in some regions gives rise to the secondary structure of the protein. This can lead to myriad serious health problems such as breathlessness, dizziness, headaches, and abdominal pain for those affected by this disease. (credit: modification of work by Ed Uthman scale-bar data from Matt Russell)īecause of this change of one amino acid in the chain, hemoglobin molecules form long fibers that distort the biconcave, or disc-shaped, red blood cells and assume a crescent or “sickle” shape, which clogs arteries (Figure 3). In this blood smear, visualized at 535x magnification using bright field microscopy, sickle cells are crescent shaped, while normal cells are disc-shaped. What is even more remarkable is that those 600 amino acids are encoded by three nucleotides each, and the mutation is caused by a single base change (point mutation), 1 in 1800 bases.įigure 3. The structural difference between a normal hemoglobin molecule and a sickle cell molecule-which dramatically decreases life expectancy-is a single amino acid of the 600. The molecule, therefore, has about 600 amino acids. What is most remarkable to consider is that a hemoglobin molecule is made up of two alpha chains and two beta chains that each consist of about 150 amino acids. Specifically, the amino acid glutamic acid is substituted by valine in the β chain. In sickle cell anemia, the hemoglobin β chain (a small portion of which is shown in Figure 2) has a single amino acid substitution, causing a change in protein structure and function. A change in nucleotide sequence of the gene’s coding region may lead to a different amino acid being added to the growing polypeptide chain, causing a change in protein structure and function. The unique sequence for every protein is ultimately determined by the gene encoding the protein. In sickle cell hemoglobin, this glutamate is replaced by a valine. In normal hemoglobin, the amino acid at position seven is glutamate. The beta chain of hemoglobin is 147 residues in length, yet a single amino acid substitution leads to sickle cell anemia. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
